Clinical Grading Scales for Predicting Early Neurological Worsening in Spontaneous Intracerebral Hemorrhage
Luca Masotti, Gianni Lorenzini, Paolo Pennati, Daniel Agustin Godoy, Aurel Popa-Wagner, J Claude Hemphill, Adrian R Parry- Jones, Réza Behrouz, Natalia S Rost and Mario Di Napoli
1Internal Medicine, Cecina Hospital, Cecina, Livorno, and Santa Maria Nuova Hospital, Florence, Italy
2Department of Medicine and Medical Specialties, Florence, Italy
3Emergency Department, Cecina Hospital, Cecina, Italy
4Neurocritical Care Unit, Sanatorio Pasteur, and Intensive Care Unit, San Juan Bautista Hospital, Catamarca, Argentina
5Molecular Psychiatry, Department of Psychiatry, Medical University Rostock, Germany
6Department of Neurology, San Francisco General Hospital, University of California, San Francisco, California, US
7The University of Manchester, Manchester Academic Health Sciences Centre, Salford Royal NHS Foundation Trust, UK
8Division of Cerebrovascular Diseases, Department of Neurology, School of Medicine, University of Texas Health Science Center, San Antonio, TX, USA
9Stroke Division, Department of Neurology, Massachusetts General Hospital, Harvard Medical School, Boston, Massachusetts, US
10Neurological Service, San Camillo de’ Lellis General Hospital, Rieti, Italy
11Neurological Section-Neuroepidemiological Unit, SMDNCenter for Cardiovascular Medicine and Cerebrovascular Disease Prevention, Sulmona, L’Aquila, Italy
- Corresponding Author:
- Luca Masotti
MD Internal Medicine
Santa Maria Nuova Hospital, Florence, Italy
E-mail: luca.masotti@tin.it
Received Date: January 30, 2016; Accepted Date: March 07, 2016; Published Date: March 13, 2016
Citation: Masotti L, Lorenzini G, Pennati P, et al. Clinical Grading Scales for Predicting Early Neurological Worsening in Spontaneous Intracerebral Hemorrhage . Stroke Res Ther. 2016, 1:1.
Abstract
Clinical Grading Scales for Predicting Early Neurological Worsening in Spontaneous Intracerebral Hemorrhage
Introduction: Early neurological worsening (ENW) is a major determinant of death in spontaneous intracerebral hemorrhage. In recent years, several clinical grading scales have been developed to identify patients at risk of 30-day or 90-day poor outcome following spontaneous (ICH), but whether these are able to predict ENW remains unclear. The aim of our study was to validate and compare two ICH grading scales for predicting ENW.
Methods: Original (o) ICH and FUNC scores were calculated for 128 consecutive, prospectively-collected ICH patients using components and cutoff values identical to those described in the original derivation cohorts. Receiver operating characteristic (ROC) analysis, including area under the curve (AUC), was used to assess the ability of each score to predict ENW.
Results: Overall, thirty-day mortality rate was 33.6% (n=43), while only 19 (15.3%) patients had good outcome at 90-days. ENW was evident in 40 patients (31.3%). Compared with patients without ENW, patients with ENW had significantly higher in-hospital mortality (79.1% vs 5%, p=0.0001), mortality within 48 hours (56.5% vs 0%,p=0.0001), baseline hematoma volume greater than 30 mm3(85.4% vs 20%, p=0.0001), baseline intraventricular bleeding (83% vs 16.25%, p=0.0001), hematoma enlargement in second brain CT (34% vs 2.5%, p=0.0001),history of vitamin K antagonists or antiplatelet treatment (75% vs 46.25%, p=0.001), lower median Glasgow Coma Scale at baseline (9.5 vs 15, p=0.001), higher median oICH score at baseline (4 vs 1, p=0.001), lower FUNC score at baseline (4.5 vs 8, p=0.001). Both scores were accurate predictors of study outcomes demonstrating excellent discrimination (AUC>0.80). However, oICH score permitted a correct ENW risk reclassification in 26% of patients.
Conclusions: oICH and FUNC scores are appropriate tools to assess ENW, with potential applications to clinical practice and research.
Keywords
Early deterioration; Score; Intracerebral hemorrhage; Outcome; Prognosis
Background
Spontaneous intracerebral hemorrhage (ICH) is a devastating illness with limited treatments [1]. There is a robust theoretical and experimental basis to support aggressive management of acute ICH, but the role of neurosurgical interventions remains controversial and has not shown clear benefit over standard medical care [2-5].
Deciding the appropriate level of medical care to offer ICH patients in the acute phase can be difficult and approaches vary likely influenced by the views of the clinician and patient (or the patient’s next of kin) regarding prognosis. Despite the availability of validated prognostic scores, such tools remain underutilized, in part because of their multiplicity [6-8]. However, there are several potential benefits of prognostic scores. First, they may aid physicians in making difficult decisions regarding the use of intensive care resources and other aggressive medical care, or help with planning end of life care to facilitate a dignified and peaceful death. Second, patients and their next of kin should be provided with an accurate, unbiased prognosis and a reliable model for outcome in ICH can help to achieve this. Third, prognostic scores are of interest to hospitals and clinicians for quality assurance, allowing comparison of outcomes between providers and monitoring of risk-adjusted outcomes for single providers overtime [9]. Hemphill’s original ICH score (oICH) and FUNC score are perhaps the best known and most extensively used scores worldwide [10,11]. Both are derived from North American populations, and allow estimation of the absolute risk of 30-day mortality (oICH) and 90-day functional dependence (FUNC). External validation studies have largely focused on the oICH score (6) but a few studies have validated the FUNC score only in North American populations [12,13]. Early neurological worsening (ENW) represents a major determinant of poor outcome in patients suffering for [14]. Much recently a systematic review in which ENW was defined as death or worsening on a reliable outcome scale within seven days from ICH onset, found that elevated blood glucose and d-Dimer levels, ICH volume and intraventricular extension of bleeding were significant risk factors for ENW [14].
Prediction of ENW was not examined in the derivation cohorts for the oICH and FUNC scores, therefore our aim was to test the hypothesis that the oICH and FUNC scores would predict ENW and to compare their performance in an unselected sample of consecutive ICH patients.
Materials and Methods
We studied all patients admitted with a diagnosis of sICH within 24 hours after stroke onset, within the calendar years 2006 to 2012 in a primary Italian hospital. All sICH patients with a baseline admission CT scan and data available on 30-day mortality and 90- day functional status were considered eligible. sICH was defined as the sudden occurrence of bleeding into the cerebral parenchyma, with or without intraventricular extension, confirmed by the clinical picture and CT scan, in absence of trauma, surgery or clear evidence of an underlying structural cause. Details on study methodology have been previously described [15,16]. All CT scans were downloaded directly to a workstation and stored in DICOM format where they were reviewed by study investigators blinded to clinical data. Neuroradiological findings were classified according to localization (supratentorial or infratentorial), site of sICH, volume of hematoma, midline shift, intraventricular extent of hemorrhage, and presence of hydrocephalus. ICH was considered lobar in location if the origin of the hemorrhage appeared to be in the cerebral hemispheres superficial to the deep gray matter structures. Hemorrhages originating in the thalamus and basal ganglia were considered “deep” in location. ICH hematoma volume was measured using ABC/2 method [17]. All variables were determined on the initial CT scan.
Clinical variables, including hypertension, diabetes, and coronary artery disease, were defined as previously reported [15,16]. Glasgow Coma Scale (GCS) was defined as the first recorded on hospital admission, after resuscitation and clinical stabilization. Pre- sICH cognitive impairment was defined as a history of cognitive impairment based on family interview and medical record review. Determination of oICH and FUNC scores were performed in an identical manner to that reported in the original manuscripts (Appendix 1) [10,11]. All patients were treated according to international guidelines [18,19]. Aggressive full care during the first 24 h after ICH onset without restriction of optimal care for do-not-resuscitate (DNR) patients was assured.
Primary outcome was ENW, defined as ≥3 point decrease in the Glasgow Coma Scale (GCS) score for non-comatose patients (GCS>8), or ≥2 point decrease for comatose patients (GCS ≤ 8), or the presence of a new focal neurological deficit, or worsening of previous deficit, assessed using a standardized neurological examination, or the appearance of clinical signs of brain herniation within 24 hours after ICH. Secondary outcomes were 30-day global mortality and 90-day good functional outcome (GFO), defined as Glasgow Outcome Scale (GOS) ≥ 4 [20].
All outcome events were confirmed by a study physician. We were able to obtain complete information on all included patients. Specific ethical approval was not required for this analysis as no new data collection was performed and the use of existing data for subsequent secondary analyses had been previously approved [15,16].
Statistical Analysis
Continuous variables are described as mean ± SD or median values with 25th and 75th percentiles, according to manner of distribution. Frequencies and percentages were calculated for categorical variables. The distributions of baseline risk factors in derivation and validation populations were compared using chi square test and one-way ANOVA or independent sample t-test for categorical and continuous variables, respectively.
Receiver operating characteristic (ROC) curves were constructed for each study endpoint in order to examine the predictive performance of each schema. Areas under the curve (AUC) or c-indexes were calculated for each of the study endpoints. Areas under the curve were compared using the non-parametric method [21]. CIs were constructed using DeLong’s variance estimate. Net reclassification improvement (NRI) was calculated as an index of inferior/superior performance of both scores [22]. NRI was calculated by assessing risk scores as low vs. intermediate vs. high [22,23].
Significance was indicated by a p value <0.05. Statistical analysis was performed with SPSS version 17.0 (SPSS Inc., Chicago, IL) and the R statistical package (www.r-project.org).
Results
Between November 1, 2006 and October 31, 2012, 128 consecutive patients were included. ENW occurred in 40 patients (31.3%), 73 patients (57.0%) remained stable and 15 (11.7%) improved during the first 24 hours. Case fatality rate was 19.5% (n=25) at 24 hours, 28.9% (n=37) at 7 days, 33.6% (n=43) at 30 days. At 90 days, 19 (14.8%) had a good outcome, 61 (47.7%) were alive with significant impairment, and 48 (37.5%) died. The demographic and clinical characteristics of the study population are summarized in Table 1.
| All Patients, No.=128 |
||
| Age, y (SD) | 80.1 | (9.5) |
| Male sex, n (%) | 61 | (47.7) |
| Vascular risk factors | ||
| Arterial hypertension, n (%) | 92 | (71.9) |
| Diabetes mellitus, n (%) | 22 | (17.2) |
| Hypercholesterolemia(>200 mg/dl), n (%) | 17 | (13.3) |
| Atrial fibrillation, n (%) | 28 | (21.9) |
| Congestive heart failure, n (%) | 5 | (3.9) |
| Coronary heart disease, n (%) | 17 | (13.3) |
| Previous cerebrovascular disease, (%) | 25 | (19.5) |
| Renal failure, n (%) | 10 | (7.8) |
| Cognitive impairment, n (%) | 19 | (14.8) |
| VKA treatment, n (%) | 25 | (19.5) |
| Antiplatelet treatment, n (%) | 47 | (36.7) |
| Clinical characteristics | ||
| GCS, median (25th-75th) | 14 | (4 -15) |
| oICH score, median (25th-75th) | 2 | (1-3) |
| FUNC score, median (25th-75th) | 7 | (5-9) |
| Radiological variables | ||
| Localization of the ICH, n (%) | ||
| – Nucleo-capsular | 55 | (43.0) |
| – Thalamic | 15 | (11.7) |
| – Lobar | 46 | (35.9) |
| – Cerebellar | 7 | (5.5) |
| – Pontine and brainstem | 4 | (3.1) |
| – Other | 1 | (0.8) |
| Hematoma volume | ||
| – < 30 mm3, n (%) | 71 | (55.5) |
| – 30-60 mm3, n (%) | 15 | (11.7) |
| – >60 mm3, n (%) | 42 | (32.8) |
| IVH | 53 | (41.4) |
| Outcomes | ||
| Length of in-hospital stay, days median (25th-75th) | 10 | (3-15) |
| 30-day mortality, n (%) | 43 | (33.6) |
| ENW, n (%) | 40 | (31.3) |
| 90-day GOS, median (25th-75th) | 3 | (1-3) |
Table 1: Baseline characteristics.
Compared with patients without ENW, patients with ENW had significantly higher in-hospital mortality (79.1% vs 5%, p=0.0001), mortality within 48 hours (56.5% vs 0%,p=0.0001), baseline hematoma volume greater than 30 (85.4% vs 20%, p=0.0001), baseline intraventricular bleeding (83% vs 16.25%, p=0.0001), hematoma enlargement in second brain CT (34% vs 2.5%, p=0.0001),history of vitamin K antagonists or antiplatelet treatment (75% vs 46.25%, p=0.001), lower median GCS at baseline (9.5 vs 15, p=0.001), higher median oICH score at baseline (4 vs 1, p=0.001), lower FUNC score at baseline (4.5 vs 8, p=0.001). Table 2 summarizes difference between patients with and without ENW.
| ENW | no ENW | p | |
|---|---|---|---|
| Number | 40 | 88 | |
| Female sex | 62.5% | 48.5% | 0.14 |
| Mean Age ± SDÃÆââ¬Å¡ÃâàÃÆââ¬Å¡ÃâàÃÆââ¬Å¡ÃâàÃÆââ¬Å¡Ãâà(years) | 83 ± 6 | 79 ± 11 | 0.85 |
| In-hospital mortality | 79.1% | 5% | 0.0001 |
| Mortality within 48 hours | 56.5% | 0% | 0.0001 |
| Baseline hematoma volume > 30 mm3 | 85.4% | 20% | 0.0001 |
| Baseline hematoma volume > 60 mm3 | 72.9% | 8.7% | 0.0001 |
| Baseline intraventricular bleeding | 83% | 16.25% | 0.0001 |
| Baseline midline shift | 75% | 10% | 0.0001 |
| Hematoma localization | |||
| Deep | 42% | 63.75% | 0.028 |
| LobarÃÆââ¬Å¡ÃâàÃÆââ¬Å¡ÃâàÃÆââ¬Å¡ÃâàÃÆââ¬Å¡ÃâàÃÆââ¬Å¡Ãâà| 42% | 30% | 0.26 |
| Infratentorial | 6% | 6.25% | 1.000 |
| Hematoma enlargement at second brain CT | 34% | 2.5% | 0.0001 |
| VKA or antiplatelet treatment | 75% | 46.25%ÃÆââ¬Å¡ÃâàÃÆââ¬Å¡Ãâà| 0.001 |
| Arterial hypertension | 75% | 72.5% | 0.83 |
| Cognitive impairment | 14.5% | 15% | 0.78 |
| Median* systolic blood pressure at baseline (mmHg) | 170 (151-187) | 170 (140-180) | 1.000 |
| Baseline median GCS | 9.5 (7-12.25) | 15 (14-15) | 0.0001 |
| Baseline median oICH score | 4 (3-4) | 1 (0-2) | 0.0001 |
| Baseline median FUNC score | 4.5 (3-5) | 8 (7-9) | 0.0001 |
| 90-day GOS ≤ 3 | 100% | 76.25% | 0.0001 |
Table 2: Characteristics of patients with ENW and without ENW.
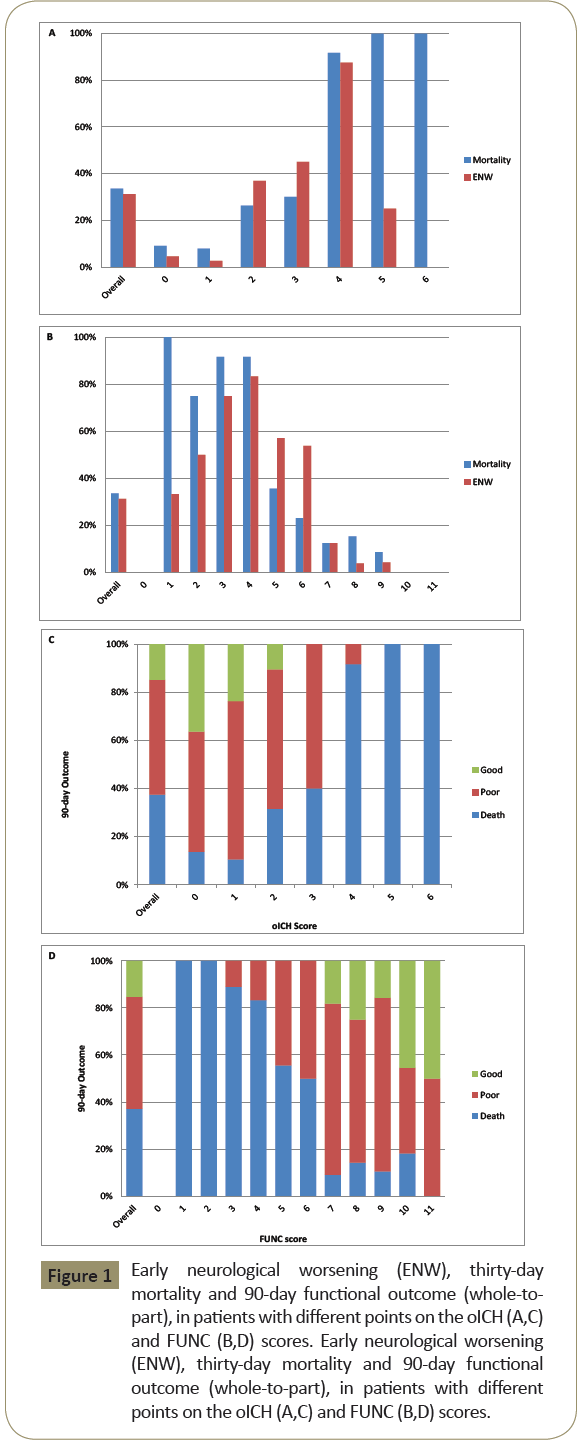
The median oICH score was 2 [interquartile range (IQR): 1-3] and it was an accurate predictor of ENW (P<0.0001; Cuzick’s test for trend) (Figure 1A). ENW occurred most often in those with an oICH score of 4 (84.5%). Similarly, oICH score was an accurate predictor of 30-day mortality (P<0.0001; Cuzick’s test for trend). Thirty-day mortality rates ranged between 9.1% for patients with a score of 0 to 100% for patients with scores ≥5, with a marked increase in mortality for a score of 4, relative to those scoring 3 (Figure1B). The distribution of 90-day outcome by oICH score is shown in the Figure 1C. With an oICH score ≥4, mortality was high and no survivors had a GFO. In logistic regression analysis, a 1-point increase in the oICH score was associated with a 3-fold increase in the odds of ENW and death, together with a reduction of 30% for the probability of GFO at 90 days (Table 3).
Figure 1: Early neurological worsening (ENW), thirty-day mortality and 90-day functional outcome (whole-topart), in patients with different points on the oICH (A,C) and FUNC (B,D) scores. Early neurological worsening (ENW), thirty-day mortality and 90-day functional outcome (whole-to-part), in patients with different points on the oICH (A,C) and FUNC (B,D) scores.
| Mortality | GFO | ENW | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Score | OR* | 95% CI | α† | β‡ | P- value | AUC | 95% CI | α† | β‡ | P- value | AUC | 95% CI | α† | β‡ | P- value | |||
| oIC | 3.3 | 2.2 | to | - | +1.22 | <0.00 | 0.34 | 0.1 | to | - | - | 0.000 | 2.69 | 1.8 | to | -3.118 | +0.99 | <0.00 |
| H | 9 | 6 | 5.07 | 3.544 | 0 | 01 | 8 | 0.61 | 0.317 | 1.087 | 4 | 9 | 3.83 | 0 | 01 | |||
| FUN | 0.4 | 0.3 | To | 3.93 | - | <0.00 | 2.08 | 1.4 | to | 7.48 | +0.73 | 0.000 | 0.53 | 0.4 | to | 3.106 | - | <0.00 |
| C | 7 | 6 | 0.60 | 9 | 0.760 | 01 | 0 | 3.10 | 9 | 2 | 3 | 3 | 0.67 | 0.629 | 01 | |||
In logit function, †α indicates slope; ‡β coefficient. Logit function is calculated according to the formula logit(P) = Χ
X indicates score punctuation.
Table 3: Logistic regression analysis of oICH and FUNC score for the outcome of 30-day mortality, good functional outcome (GFO) at 90-days, and early neurological worsening (ENW).
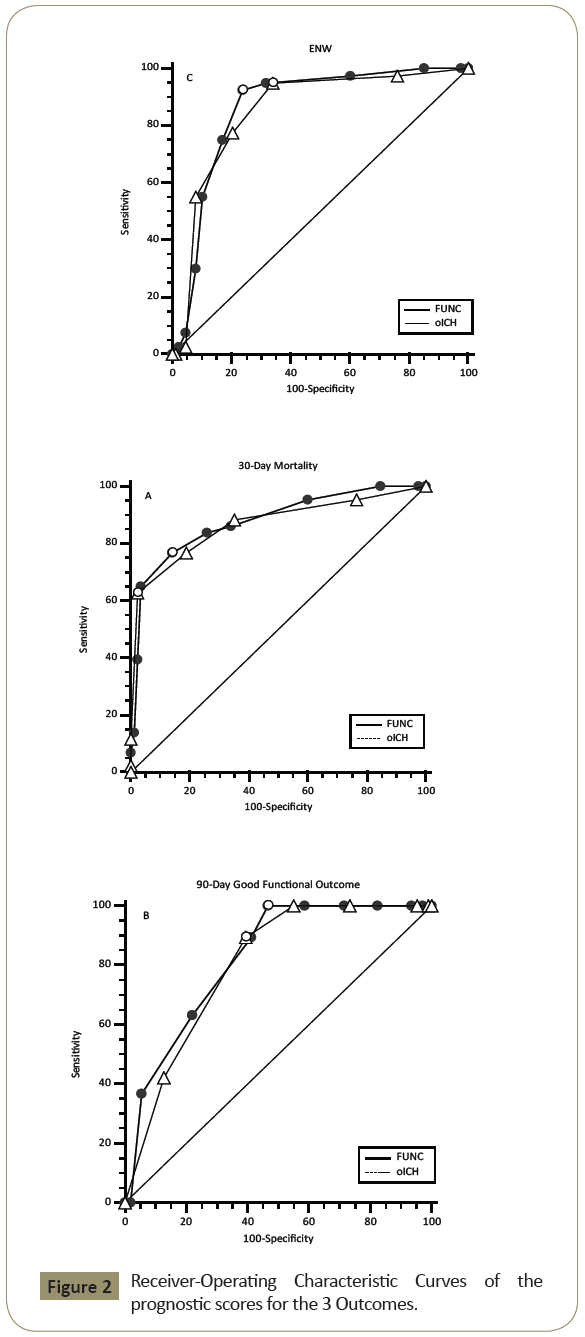
The median FUNC score was 7 (IQR: 5 to 9) and it was also an accurate predictor of ENW and 30-day mortality (P<0.0001; Cuzick’s test for trend). ENW was most likely with a FUNC score of 4 (83.3%). No patient with a FUNC score > 10 died (n=13) but each decrease in score was associated with an increase in mortality (Figure 1b). No patients with a score ≤ 6 had a GFO. Figure 1D shows the distribution of different outcomes by FUNC score. In logistic regression analysis, an increase of 1 point in the FUNC score was significantly associated with an approximate 50% reduced odds for both 30-day mortality and ENW, together with a 2-fold increased odds of a GFO at 90 days (Table 3). Both scores showed excellent discrimination, with C-statistics over 0.80 in predicting ENW and 30-day mortality (Figure 2) with a comparable prediction (P=0.5609 and P=0.5634, respectively), whereas no difference (P=0.4884) was found in prediction of GFO between the oICH score and FUNC score (Table 4).
| oICH | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| FUNC | Mortality (n=43) | GFO+ (n=19) | ENW+ (n=40) | |||||||||
| FrequencyÃÆââ¬Å¡Ãâàrowpercentage(%) | Low | Middle | High | Total | Low | Middle | High | Total | Low | Middle | High | Total |
| Low | 2 (100) | - | - | 2 | 11 (91.7) | 1 (8.3) | - | 12 | 1 (100) | - | - | 1 |
| Middle | 3 (23.1) | 7 (53.8) | 3 (23.1) | 13 | 6 (85.7) | 1 (14.3) | - | 7 | 1 (5.9) | 12 (70.6) | 4 (23.5) | 17 |
| High | - | 4 (14.3) | 24 (85.7) | 28 | - | - | - | - | - | 4 (18.2) | 18 (81.8) | 22 |
| Total | 5 | 11 | 27 | 43 | 17 | 2 | - | 19 | 2 | 16 | 22 | 40 |
| Up, n (%) | 3 (6.98) | 1 (5.26) | 4 (10.0) | |||||||||
| Down, n (%) | 7 (16.28) | 6 (31.58) | 5 (12.5) | |||||||||
| Alive (n=85) | GFO- (n=109) | ENW- (n=88) | ||||||||||
| Frequencyrow percentage (%) | Low | Middle | High | Total | Low | Middle | High | Total | Low | Middle | High | Total |
| Low | 32 (94.2) | 2 (5.8) | - | 34 | 23 (95.8) | 1 (4.2) | - | 24 | 33 (94.3) | 2 (5.7) | - | 35 |
| Middle | 23 (47.9) | 23 (47.9) | 2 (4.2) | 48 | 20 (37.0) | 29 (53.7) | 5 (9.3) | 54 | 25 (56.8) | 18 (40.9) | 1 (2.3) | 44 |
| High | - | 3 (100) | - | 3 | - | 7 (22.6) | 24 (77.4) | 31 | - | 3 (33.3) | 6 (66.7) | 9 |
| Total | 55 | 28 | 2 | 85 | 43 | 37 | 29 | 109 | 58 | 23 | 7 | 88 |
| Up, n (%) | 4 (4.71) | 4 (3.67) | 3 (3.41) | |||||||||
| Down, n (%) | 26 (30.59) | 27(24.77) | 28 (31.82) | |||||||||
| NRI* | 16.58 (z=1.6957; P=0.0899) | -5.22 (z=-0.3516; P=0.7251) | 25.91 (z=2.6405; P=0.0083) | |||||||||
NRT = [P(up \ D =1) - P(down \ D =1)]-[P(up \ D = 0) - P(down \ D = 0)]
D denotes the event indicator (1 = Dead, 0 = Alive). Upward movement (up) is a change into higher category based on the new model and downward movement (down) as a change in the opposite side.
Table 4: Reclassification tables and net reclassification improvement (NRI) for 30-day mortality, 90-day good functional outcome (GFO) and early neurological worsening (ENW) estimated by oICH and FUNC scores.
By using the oICH score in ENW risk classification, approximately 31% of individuals moved up or down a risk category with an overall net reclassification index (NRI) of about 26% when ENW indicates Early Neurological Worsening; FUNC-Rost’s Score; GCSGlasgow Coma Scale; GOS-Glasgow Outcome Scale; ICH-Intra Cerebral Hemorrhage; IVH-Intra Ventricular Hemorrhage; mRS-modified Rankin Scale; oICH-original Hemphill’s ICH Score; SD-Standard Deviation, VKAVitamin K Antagonist
compared to FUNC score (Table 5). There were no significant improvements in risk classification among both scores with respect to the definition of low, intermediate, high risk of 30-day mortality and GFO (Table 5).
| oICH score | FUNC score | |||||
|---|---|---|---|---|---|---|
| ENW | Mortality | GFO | ENW | Mortality | GFO | |
| Cut-off | 1 | 4 | 2 | 7 | 5 | 6 |
| AUC (95% CI) |
0.851 (0.781-0.921) |
0.868 (0.795-0.941) |
0.799 (0.720-0.878) |
0.862 (0.796-0.927) |
0.879 (0.813-0.945) |
0.821 (0.740-0.901) |
| Sensitivity | 0.950 | 0.976 | 0.895 | 0.925 | 0.859 | 1.0 |
| Specificity | 0.659 | 0.628 | 0.606 | 0.761 | 0.767 | 0.532 |
| +LR | 2.79 | 2.62 | 227 | 3.88 | 3.69 | 2.14 |
| -LR | 0.08 | 0.04 | 0.17 | 0.10 | 0.18 | 0.0 |
| PV+ | 0.54 | 0.53 | 0.49 | 0.62 | 0.61 | 0.48 |
| PV- | 0.97 | 0.98 | 0.93 | 0.96 | 0.93 | 1.0 |
| J index | 0.609 | 0.604 | 0.5 | 0.686 | 0.626 | 0.532 |
Table 5: Areas under the ROC curves (AUC), sensitivity, specificity, positive likelihood ratio (+LR), negative likelihood ratio (-LR), positive predictive value (PV+), and negative predictive value (PV-) of the original ICH score and FUNC score for ENW, 30-day mortality and good functional outcome (GFO) at 90-days, respectively, with the different cut-off values to achieve the best Youden’s(J) Index.
Discussion
Predicting outcomes is of utmost importance for management of spontaneous ICH. In the present study, we compared the performance of two well-known and validated ICH prognostic scales (oICH and FUNC scores) in predicting ENW, 30-day mortality and 90-day GFO in a prospectively recruited European cohort from a primary care center. We found no evidence to prefer one over the other, as both showed excellent discrimination in predicting ENW, mortality and are less effective for predicting GFO. Overall, oICH outperforms the FUNC score in predicting ENW, showing a better re-classification of patients at risk for ENW.
This comparison is of practical importance because ICH grading scales have considerable potential to help in the management of ICH patients, especially in the first hours from symptoms onset. Although experienced clinicians could predict outcomes better than scores, both FUNC and ICH scores are simple for clinicians to use, also without neurological expertise, incorporate intuitive information that is part of standard clinical and radiological bedside assessment, and offer objective standardized risk stratification.
The oICH score was developed to risk stratify patients at admission with regard to 30-day mortality [10]. The score ranges from 0 to 6. In the original cohort, a score of 3 and above was associated with significant increase in the risk of 30-day mortality (72 vs.26% for oICH score of 2) [10]. A much recent meta-analysis enclosing nine studies confirmed that oICH has a good prognostic ability to predict 30-days mortality in patients suffering for ICH (AUC 0.80, 95% CI: 0.77-0.85) [24].
The FUNC Score was intended for use during initial evaluation to predict functional independence (GOS≥4) at 3 months [12]. In the original cohort, FUNC score accurately predicted functional independence. Among patients who scored 11, more than 70% demonstrated functional independence at 90 days whereas only 45% of patients with a score of 5 achieved the same status [11].
Although the oICH and FUNC scores were not originally developed for predicting ENW, both exhibited excellent discriminatory capacity for ENW, as reflected by c-indexes greater than 0.80. Our findings see consistent with much recent literature evidence from findings of the INTERACT II trial. In fact, a sub-analysis of this trial showed that clinical grading scales such as oICH could have a greater predictive power when performed after 24-hours from ICH onset, strongly suggesting that ENW represent the instability of the ICH patients and may help to explain the variances of outcome that could not be explained by baseline oICH score [25].
The difference in score performances could be explained by local or regional differences in practice. Any restriction of care could result in larger differences in outcome compared to patients treated at centers with a more aggressive approach toward ICH, where do-not-resuscitate (DNR) orders do not impact on the level of supportive medical care provided [26]. Alternatively, early DNR orders could have a greater impact in centers in which these orders translate into limitations in the provision of standard medical therapy, such as control of hypertension, reversal of anticoagulation, and the prevention and treatment of systemic complications.
The c-index is not the only parameter to be taken into consideration in determining the utility or predictive ability of risk estimation schemas [27] From the clinical perspective, reclassification analysis is more important, especially for patients reclassified correctly from low and intermediate risk to high risk, as these patients will be considered for alternative treatment. The oICH was significantly better than the FUNC score in predicting ENW. The oICH score correctly reclassified about one quarter of patients for ENW risk. The fact that the oICH was predictive of ENW is an advantage, as ENW risk prediction alerts the physician of a high risk of acute complications and thus may help to stratify the patient for certain therapies, such as, ultra-early hemostatic therapy or surgery.
An early and accurate estimate of prognosis after ICH is likely to be welcomed by patients and their families and may be useful to physicians in planning patient care. Furthermore, as healthcare costs have increased, outcome assessment has become a major priority. Accurate and objective outcome prediction will allow for financial and human resources to be allocated appropriately. Finally, a prognostic score allows comparison of observed versus predicted outcomes. The difference between actual and predicted outcomes may provide a benchmark of quality of care and facilitate insight into means for improving performance.
We recognize that our study presents limitations. Our inability to demonstrate a significant difference between the two scores could be a potential limitation of this study, likely due to a relatively small sample size. However, the similarity in performance between the two scores suggests that these grading scales can both be used, the choice depending on local preferences. For example, the FUNC score may be more useful in predicting long-term outcomes, so it may be preferred if this is a priority. Although sample size was insufficient to establish the relative usefulness of each scale, the J Index, sensitivity, specificity, positive predictive value, and negative predictive value offer useful information about the relative strengths and weaknesses of each scale. However, the results of relatively novel performance measures suggest the oICH may be preferred.
Prediction of ENW was not examined in the derivation cohorts for the oICH and FUNC scores, but was studied in our cohort. Most cases of neurological deterioration after ICH occur early and are often fatal [28]. Both scores represent sensitive and specific tools for the identification of ENW, as defined in our study. We recognize that the definitions of ENW vary, making it difficult to compare studies [29]. We have used a decrease in the GCS for non-comatose patients of ≥3 points and of ≥ 2 points for comatose patients since worse outcome has been demonstrated [29-32]. A prospective study to determine the relative efficiency and cost-effectiveness of this strategy for the evaluation of ENW with historical data is planned.
Conclusion
In conclusion, the results of our study suggest that the oICH and FUNC scores are substantially equivalent, as both have excellent discrimination for ENW other than GFO at 90-days and 30-day mortality. Both are appropriate tools to assess severity of ICH, with applications to clinical practice and research.
References
- Anderson CS, Heeley E, Huang Y, Wang J, Stapf C, et al. (2013) Rapid blood-pressure lowering in patients with acute intracerebral hemorrhage. N Engl J Med 368:2355-2365.
- Balami JS, Buchan AM (2012) Complications of intracerebral haemorrhage. Lancet Neurol11:101-118.
- Elliott J, Smith M (2010) The acute management of intracerebral hemorrhage: a clinical review. AnesthAnalg110:1419-1427.
- Gregson BA, Broderick JP, Auer LM, Batjer H, Chen XC, et al. (2012) Individual patient data subgroup meta-analysis of surgery for spontaneous supratentorial intracerebral hemorrhage. Stroke 43:1496-1504.
- Mendelow AD, Gregson BA, Rowan EN, Murray GD, Gholkar A, et al. (2013) Early surgery versus initial conservative treatment in patients with spontaneous supratentorial lobar intracerebral haematomas (STICH II): a randomised trial. Lancet 382:397-408.
- Bruce SS, Appelboom G, Piazza M, Hwang BY, Kellner C, et al. (2011) A Comparative Evaluation of Existing Grading Scales inIntracerebral Hemorrhage. Neurocrit Care 15: 498-505.
- Hwang BY, Appelboom G, Kellner CP, Carpenter AM, Kellner MA, et al. (2010) Clinical grading scales in intracerebral hemorrhage. Neurocrit Care 13:141-151.
- Di Napoli M (2011) Scale prognostichenell'emorragiacerebrale. Reviews in Health Care 2:69-76.
- Godoy DA, Pinero G, Di Napoli M (2006) Predicting mortality in spontaneous intracerebral hemorrhage:can modification to original score improve the prediction? Stroke37:1038-1044.
- Hemphill JC 3rd, Bonovich DC, Besmertis L, Manley GT, Johnston SC (2001) The ICH score: a simple, reliable grading scale for intracerebral hemorrhage. Stroke 32:891-897.
- Rost NS, Smith EE, Chang Y, Snider RW, Chanderraj R, et al. (2008) Prediction of functional outcome in patients with primary intracerebral hemorrhage: the FUNC score. Stroke 39:2304-2309.
- Garrett JS, Zarghouni M, Layton KF, Graybeal D, Daoud YA (2013) Validation of Clinical Prediction Scores in Patients withPrimary Intracerebral Hemorrhage. Neurocrit Care 19:329-335.
- Mittal MK, Lele A (2011) Predictors of poor outcome at hospital discharge following a spontaneous intracerebral hemorrhage. Int J Neurosci121:267-270.
- Specogna AV, Turin TC, Patten SB, Hill MD (2014) Factors associated with early deterioration after spontaneous intracerebral hemorrhage: a systematic review and meta-analysis. PLOS one 9: e96743.
- Di Napoli M, Godoy DA, Campi V, del Valle M, Piñero G, et al. (2011) C-reactive protein level measurement improves mortality prediction when added to the spontaneous intracerebral hemorrhage score. Stroke 42:1230-1236.
- Di Napoli M, Godoy DA, Campi V, Masotti L, Smith CJ, et al. (2012) C-reactive protein in intracerebral hemorrhage: time course, tissue localization, and prognosis. Neurology 79:690-699.
- Teasdale G, Jennett B (1974) Assessment of coma and impaired consciousness. A practical scale. Lancet2:81-84.
- Broderick J, Connolly S, Feldmann E, Hanley D, Kase C, et al. (2007) Guidelines for the management of spontaneous intracerebral hemorrhage in adults: 2007 update: a guideline from the American Heart Association/American Stroke Association Stroke Council, High Blood Pressure Research Council, and the Quality of Care and Outcomes in Research Interdisciplinary Working Group. Stroke 38:2001-2023.
- Hemphill JC 3rd, Greenberg SM, Anderson CS, Becker K, Bendok BR, et al. (2010) Guidelines for the management of spontaneous intracerebral hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 41:2108-2129.
- Jennett B, Snoek J, Bond MR, Brooks N (1981) Disability after severe head injury: observations on the use of the Glasgow Outcome Scale. J NeurolNeurosurg Psychiatry 44:285-293.
- DeLong ER, DeLong DM, Clarke-Pearson DL (1988) Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 44:837-845.
- Pencina MJ, D'Agostino RB, Vasan RS (2010) Statistical methods for assessment of added usefulness of new biomarkers. Clin Chem Lab Med 48:1703-1711.
- Vickers AJ, Elkin EB (2006) Decision curve analysis: a novel method for evaluating prediction models. Med Decis Making 26:565-574.
- Mattishent K, Kwok CS, Ashkir L, Pelpola K, Myint PK, et al. (2015) Prognostic Tools for Early Mortality in Hemorrhagic Stroke: Systematic Review and. Meta-Analysis. J Clin Neurol11:339-348.
- Heeley E, Anderson CS, Woodward M, Arima H, Robinson T, et al. (2015) Poor utility of grading scales in acute intracerebral hemorrhage: results from the INTERACT2 trial. Int J Stroke 10:1101-1107.
- Jain A, Jain M, Bellolio MF, Schears RM, Rabinstein AA, et al. (2013) Is early DNR a self-fulfilling prophecy for patients with spontaneous intracerebral hemorrhage? Neurocrit Care 19:342-346.
- Steyerberg EW, Vickers AJ, Cook NR, Gerds T, Gonen M, et al. (2010) Assessing the performance of prediction models: a framework for traditional and novel measures. Epidemiology 21:128-138.
- Brott T, Broderick J, Kothari R, Barsan W, Tomsick T, et al. (1997) Early hemorrhage growth in patients with intracerebral hemorrhage. Stroke 28:1-5.
- Siegler JE, Martin-Schild S (2011) Early Neurological Deterioration (END) after stroke: the END depends on the definition. Int J Stroke 6:211-212.
- Moon JS, Janjua N, Ahmed S, Kirmani JF, Harris-Lane P,et al. (2008) Prehospital neurologic deterioration in patients with intracerebral hemorrhage. Crit Care Med 36:172-175.
- Holdgate A, Ching N, Angonese L (2006) Variability in agreement between physicians and nurses when measuring the Glasgow Coma Scale in the emergency department limits its clinical usefulness. Emerg Med Australas18:379-384.
- Kornbluth J, Bhardwaj A (2011) Evaluation of coma: a critical appraisal of popular scoring systems. Neurocrit Care 14:134-143.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences