Motor and Cognitive Impairment after Stroke: A Common Bond or a Simultaneous Deficit?
Sonja Verstraeten, Ruth Mark and Margriet Sitskoorn
1 Department of Medical Psychology, Máxima Medical Center, The Netherlands
2Department of Cognitive Neuropsychology, Tilburg University, The Netherlands
- Corresponding Author:
- Sonja Verstraeten
Máxima Medical Center, Department of Medical Psychology, The Netherlands
Tel: +31 408889250
E-mail: S.Verstraeten@mmc.nl
Received Date: January 18, 2016; Accepted Date: February 19, 2016; Published Date: February 25, 2016
Citation: Verstraeten S, Mark R, Sitskoorn M, Motor and Cognitive Impairment after Stroke: A Common Bond or a Simultaneous Deficit?. Stroke Res Ther. 2016, 1:1.
Abstract
Motor and Cognitive Impairment after Stroke: A Common Bond or a Simultaneous Deficit?
Background: The prevalence of both motor deficit and cognitive impairment after stroke is high and persistent. Motor impairment, especially paresis, is often more obvious to both patients and their carers while cognitive problems can also have devastating effects on quality of life. The current review explores whether there might be a link between motor and cognitive impairment after stroke in the same patients.
Evidence acquisition: Electronic databases were searched systematically. Studies fulfilled the selection criteria when they were written in English and when both motor and cognitive deficit after stroke were evaluated and analyzed in the same patients, either with objective or subjective measures.
Results: 20 papers were included in this review. Measures of gait, balance and limb function were consistently correlated with aspects of attention and executive functioning in 9 studies. Correlations were robust when objective measures for specific functions were used, while results based on subjective or global measures tended to be more heterogeneous.
Conclusions: Motor and cognitive impairment appear to be linked in stroke. Studies which assessed correlations between the two domains in the same patients (irrespective of time post stroke) found, on the whole, strong links between walking and balance tasks and the cognitive domains of attention and executive functioning. Whether the same mechanisms or neural pathways underlie these deficits or whether they are occurring simultaneously remains to be explored.
Keywords
stroke; Motor impairment; Cognitive deficit
Introduction
Stroke often results in both motor impairment and cognitive deficit. Motor deficits are however typically more obvious than cognitive problems to patients and their carers and rehabilitation efforts often focus on attempting to improve limb function rather than on whether the patient has trouble planning their day for example. However, even when stroke is considered mild or patients show good recovery, cognitive deficits are common [1,2]. In the first month post-stroke 49 to 91% of the patients suffer cognitive impairment in at least one cognitive domain [3,4]. Frequently, these deficits are persistent, lasting for years [5]. Since cognitive impairment can affect social, professional and vocational functioning [6,7], influence quality of life of both patients and family members [8,9] and can affect rehabilitation success [10,11], it is important to assess both domains - motor and cognitive in the same patients.
The link between motor deficit and cognitive impairment has not specifically been a subject of interest in stroke research until now, while this link was made in other populations, including healthy people and patients with neurological disorders. Motor and cognitive performances were investigated simultaneously in a number of studies in healthy people. In children aged 5 to 6 years a link between specific aspects of motor and cognitive performance was found, after correcting for the influence of attention [12] and in adolescents aged 12 to 16 years an overlap between motor coordination and executive functioning (EF) was reported [13]. In older healthy people, the link between gait and cognitive functioning was studied and two main findings emerge; EF is required for both routine gait [14] and performing complex walking tasks [15]. In the oldest old population (i.e. aged 85 plus) reduced handgrip strength predicted an accelerated decline in both ADL- and global cognitive functioning, the latter evaluated using the Mini Mental State Examination (MMSE) [16].
In neurological populations, a link between motor deficit and cognitive impairment has also been reported. In Parkinson’s disease different types of motor impairment were associated with specific cognitive deficits, including deficits in attention, mental flexibility, working memory, visuospatial function and memory [17]. Furthermore, in Multiple Sclerosis measures of EF accounted for most of the variance in motor performance [18]. In three different reviews of the literature, links between gait and cognitive performance in both normal and demented populations were investigated; in both normal ageing and the most prevalent subtypes of dementia a close relationship between gait and cognition [19], more specifically attention [20] and EF [20,21] was found.
The current review attempted to explore whether there is a link between motor deficit and cognitive impairment in the same stroke patients as well as to assess which aspects of these (global) domains are linked. Our goal is to determine if there is a specific relation or pattern between motor and cognitive deficit which will help us to advise clinicians as to which cognitive domains to assess after stroke.
Materials and Methods
A systematic search of the literature between January 1980 and December 2014 was carried out using the following electronic databases: PUBMED, EMBASE, PsycINFO, PsycARTICLES and Psychology and Behavioral Science Collection. The following keywords (and combinations thereof) were used: stroke, cerebral vascular accident, cerebrovascular accident, cerebrovascular disorders or cerebral hemorrhage, cogniti*, cognitive, attention, executive function, executive control, executive function, psychomotor performance, processing speed, visuospatial function, visual perception, space perception or memory, gait*, gait-variability, walk, walking, locomotor behavior, motor activity*, motor skills, hand strength and hand grip strength. Searches were limited to human subjects and papers written in English. Reference sections were hand searched for relevant articles. Only studies which assessed and subsequently statistically evaluated both cognitive and motor performance (using specific tasks for each domain) within the same stroke patients were selected. Two reviewers (S.V., R.M.) independently assessed titles, abstracts and full-text reports on eligibility. The quality of each of the selected papers was subsequently determined by using a 13-item checklist (Table 1). Disagreement between the raters about eligibility or quality was solved by discussion and the total score each paper received was intended for descriptive purposes only.
| Each criterion was scored 1 point if the following aspects were met: |
|---|
| Study sample A. A sample of the general stroke population is taken, patients are not selected based on specific test results, age or side of lesion B. Mean or median and standard deviation or range of the time-interval after stroke is reported C. More than one clinical variable of the patient group is described (e.g. type of stroke, severity of stroke, lesion side, lesion location) D. More than one socio-demographic variable of the patient group is described (e.g. age, employment status, educational status) E. Inclusion and/or exclusion criteria are described Design F. Data is prospectively gathered G. The results are compared to at least one other non-stroke sampleÃÆââ¬Å¡Ãâà(e.g. healthy population, other patient group) H. Information is given about the selection of the patient sample (e.g. demographic and stroke characteristics of the non-participants are compared to the participants) I. At least one published standardized test is used forÃÆââ¬Å¡Ãâàthe assessment of motor functioning J. At least one published standardized test is used forÃÆââ¬Å¡Ãâàthe assessment of cognitive functioning K. Results of motor and cognitive functioning are collected at same point in time Statistical methods L. Link between motor and cognitive measures is analyzed using known statistical procedures and the results are shown in the paper M. The process of data collection is described (e.g. interview, self-report questionnaires, neuropsychological assessment) |
Table 1: List of criteria for assessing the quality of studies included.
Results
Inclusion of articles
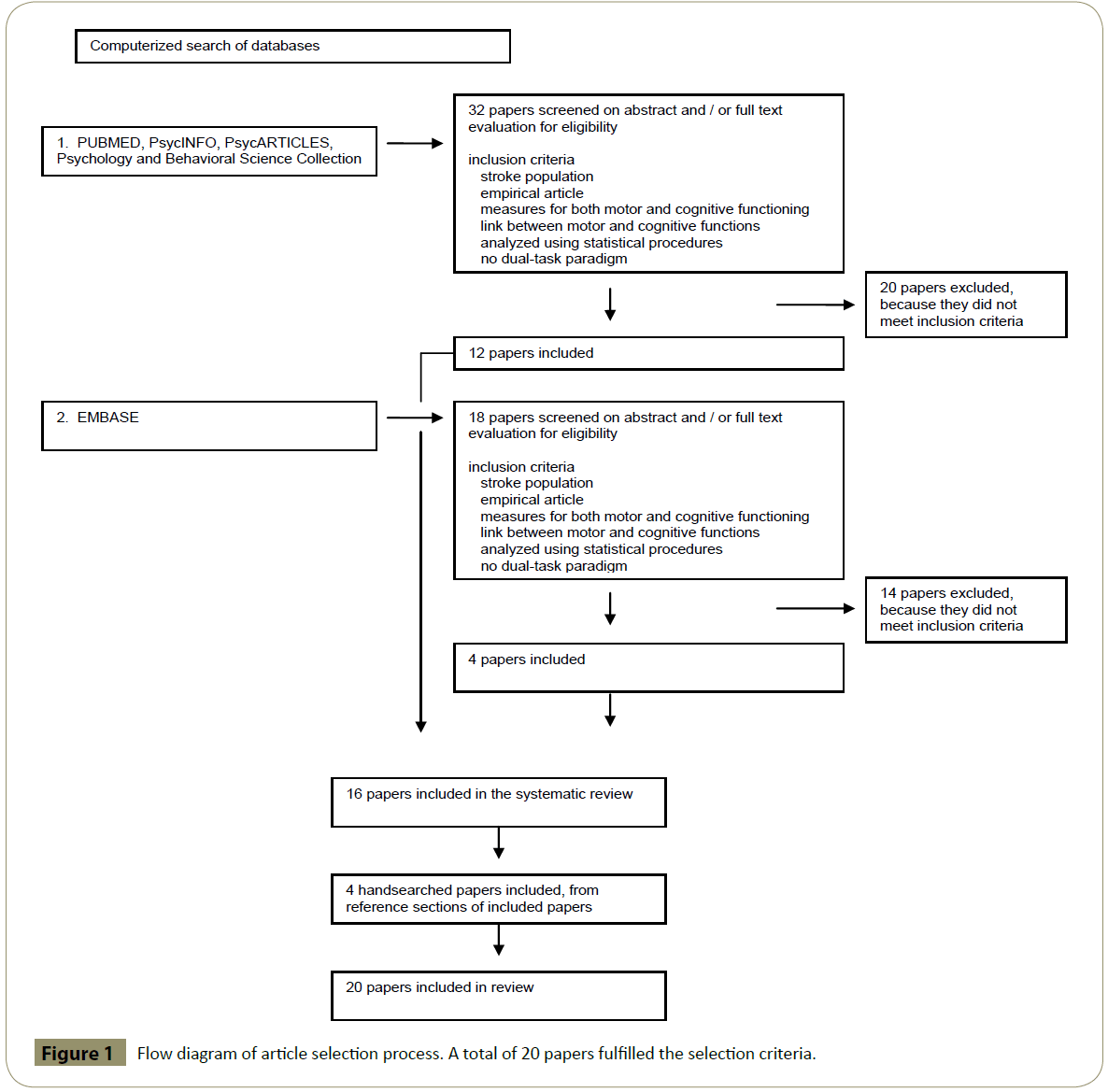
16 papers were included after searching the electronic databases, of which 2 papers were from the same authors [22,23]. However, the results come from different populations and both were therefore included. Reference sections were explored and 4 additional papers were identified. Figure 1 shows the details of the search strategy.
Figure 1: HPLC chromatogram of the nine reference compounds in 50% aqueous methanol, measured at 370nm. Retention times for rutin, sutherlandin A, sutherlandin B, kaempferol-3-O-rutinoside, sutherlandin C, sutherlandin D, quercitrin, quercetin and kaempferol were 11.9, 12.7, 13.8, 15.3, 16.2, 17.0, 18.0, 26.2 and 28.1 minutes, respectively.
General characteristics of the studies
A prospective design was employed in all of the 20 included papers. In 5 studies no specific in- or exclusion criteria were applied [24-28]. In the remaining 15 papers specific criteria were applied including, for example, age at time of stroke or minimum scores on certain motor or cognitive screening scales or tasks. Only 2 of the total 20 studies included a control group (healthy [29] or infantile hemiplegia patients [30]). The sample sizes varied from 13 to 315 participants. Mean age of the samples varied from 57.1 to 78 years. Diversity in time post stroke was substantial, ranging from 1 week to more than 6 years. In assessing motor impairment 35 different measures were used across studies; only 8 of these measures were used in more than one paper. Cognitive deficit was evaluated with 25 different tasks, 8 tasks were used in more than one study. Rating scales predominated in evaluating motor impairment. In assessing cognitive impairment objective measures were more commonly used than subjective rating scales. In 15 papers motor and cognitive impairment were evaluated at the same time point, while 5 studies used a longitudinal design and focused on predicting motor outcome. In 1 paper both types of results were reported. A summary of the 20 papers included is presented in Table 2.
| First Author | Participants |
Population |
Assessment measures | Related motor and cognitive outcome measures |
Correlation | ||||
|---|---|---|---|---|---|---|---|---|---|
N male/female age mean ± SD (range) as reported |
Side of lesion (Left /Right /Other) Time since onset mean ± SD (range) as reported |
Motor | Cognitive | Motor | Cognitive | r | |||
| Gait/locomotion | |||||||||
| Fong et al. 2001 (33) |
37 24 / 13 62.3 ± 5.4 |
22 / 15 / 0 1 wk post stroke |
- Fugl-Meyer Assessment - Functional Independence Measure (motor subscale) |
- Neurobehavioral Cognitive Status Examination - Functional Independence Measure (cognitive subscale) |
Locomotion | Comprehension | Admission 0.29* 2 weekÃÆââ¬Å¡ÃâàÃÆââ¬Å¡ÃâàÃÆââ¬Å¡ÃâàÃÆââ¬Å¡ÃâàÃÆââ¬Å¡Ãâà0.40* 4 weekÃÆââ¬Å¡ÃâàÃÆââ¬Å¡ÃâàÃÆââ¬Å¡ÃâàÃÆââ¬Å¡ÃâàÃÆââ¬Å¡Ãâà0.36* dischargeÃÆââ¬Å¡Ãâà0.52** |
||
| Hayes et al. 2012 (31) |
20 14 / 6 69.1 ± 9.4 |
9 / 11 6.89 ± 4.9 year |
- Motor Assessment Scale - Basic and complex 10 meters test |
- Trailmaking Test - Stroop Colour-Word Test - Zoo Map (BADS) - Frontal Assessment Battery - Digit Span Backwards Test |
Basic gait Basic gait |
Cognitive flexibility Working memory |
ÃÆââ¬Å¡Ãâà0.79** n.s. |
||
| Liu-Ambrose et al. 2007 (32) |
63 37 / 26 65 ± 9 (52-87) |
41 / 22 / 0 6 ± 5 year (1-28) |
- Berg Balance Scale - Timed up-and-go test - Six minutes walk test - Stair climbing time - Gait velocity - Quadriceps strength - Physical activity Scale for Individuals with Physical Disabilities |
- Stroop Colour-Word Test - Verbal Digit Span Backward Test |
Gait Gait Gait velocity Gait velocity Balance Balance |
Cognitive flexibility Working memory Cognitive flexibility Working memory Cognitive flexibility Working memory |
ÃÆââ¬Å¡Ãâà0.25* 0.22 0.25 0.22 0.34** 0.18 |
||
| Taylor-Pillae et al. 2012 (28) |
100 54 / 46 70 ± 10 |
100 Stroke 39 / 42 / 19 39 ± 49 months (3-356) |
- 4-m gait speed test - Modified Rankin Scale - 2-min step-in-place test - Chair stand test - Leg strength - Single Leg Stance Test |
- Mini Mental State Examination |
Gait velocity Balance | MMSE MMSE | ÃÆââ¬Å¡Ãâà0.20 - 0.04 | ||
| Balance | |||||||||
| Hyndman et al.ÃÆââ¬Å¡Ãâà2003ÃÆââ¬Å¡Ãâà(22) | 48 30/18 68.4 ± 11.7 |
21 / 26 / 1 46 ± 45.7 months |
- Berg Balance Scale - Nottingham Extended ADL scale - Fall events (questions) |
- Test of Everyday Attention - Star cancellation Test |
Balance Balance |
Sustained attention Divided attention |
ÃÆââ¬Å¡Ãâà0.40** 0.51** |
||
| Hyndman et al. 2008 (23) | 122 82 / 67 70.2 ± 12.5 (21-92) |
58 / 63 / 1 79.2 ± 57.3 days (10-330) |
- Berg balance scale - Rivermead Motor Assessment Scale - Nottingham Extended Activities of Daily Living Scale |
- Test of Everyday Attention |
Balance Balance Balance Balance Limb Limb Limb |
Sustained attention Divided attention1 Visual selective1 Auditory selective Sustained attention Divided attention1 Visual selective1 |
ÃÆââ¬Å¡Ãâà0.39** - 0.32** - 0.48** 0.32** 0.35** - 0.25** - 0.39** |
||
| Stapleton et al. 2012 (34) |
13 8/5 60 (21-80) |
3 / 10 / 0 median 34 days (12-129) |
- Berg Balance Scale - Semi-structured interview (falls) |
- Test of Everyday Attention - Star cancellation test |
Balance | Sustained attention Visual selective1 Auditory selective |
ÃÆââ¬Å¡Ãâà0.40 - 0.35ÃÆââ¬Å¡Ãâà0.67** |
||
| Limb function | |||||||||
| Robertson et al.
1997 (26) |
47 25 / 22 63.4 ± 12.6 |
0 / 47 / 0 25.2 ± 4.0 months |
- Barthel index - Nottingham Extended Activities of Daily Living Scale - Rivermead mobility scale - Nine-hole peg test |
- Test of Everyday Attention |
Arm function1 (dexterity of contralesional hand) |
Sustained attention Divided attention |
- 0.41** n.s |
||
| Sunderland et al. 1999 (35) |
30 21 / 9 62.5 ± 12 34 control 15 / 19 65 ± 11.5 |
15 / 15 / 0 18 days (2.5 ÃÆâÃâââ¬Ãâââ¬Å 30) |
- Dexterity in Simulations of Everyday Tasks - Jebsen Hand Function Test - Williams Doors - Grip strength |
- Apraxia Assessment - Line Cancellation - Judgement of Line Orientation - Token Test (part I and V) |
Arm function (dexterity ipsilateral hand)) | Visuospatial functioning in RCVA Action imitation in LCVA | ÃÆââ¬Å¡Ãâàn.s.* n.s.* |
||
| Sveen et al. 1999 (27) |
65 37 / 28 74.4 (56-90) |
37 / 27 / 1 1 year post stroke |
- Sodring Motor Evaluation of Stroke patients - Barthel Index - Frenchay Activities Index |
- Assessment of Stroke and other Brain damage |
Arm function Arm function |
Ideational apraxia Visuospatial constr. |
0.40† 0.44† |
||
| (subjective) Motor performance | |||||||||
| Bernspang et al. 1987 (24) |
109 69 / 40 69 ±10 |
66 / 41 / 2 within 2 weeks post stroke |
- Self-care ability assessed by principles of Fugl-Meyer and JAASKO - Motor function assessed by principles of Twitchell |
- Visual perception assessed with a 16-item bedside test - Orientation in Time and Space assessed with 5 items |
Motor function Motor function |
Visual perception Orientation in time and space |
0.05/0.08† 0.09† | ||
| Deshpande et al.1999ÃÆââ¬Å¡Ãâà(30) |
13 12 / 1 not specified |
6 / 7 / 0 > 6 months |
- Brunstrom approach |
- LOTCA Battery |
Motor perform. | Cognitive Perceptual Performance | n.s. † | ||
| Katz et al. 2000 (36) |
40 27 / 13 58 ± 9.1 |
n.a. > 6 months |
- Functional Independence Measure (motor subscale) - Rabideau Kitchen Evaluation Revised |
- LOTCA Battery |
Motor perform. | Visuomotor Organ. | Neglect AdmissionÃÆââ¬Å¡Ãâà0.48* DischargeÃÆââ¬Å¡Ãâà0.66** Non neglect AdmissionÃÆââ¬Å¡Ãâà0.26 DischargeÃÆââ¬Å¡Ãâà0.54** |
||
| Kizony et al. 2002 (38) |
30 17 /13 71.33 ± 8.39 |
7 / 23 / 0 4.7 ± 2.89 weeks |
- Assessment of Motor and Process Skills |
- Neurobehavioral Cognitive Status Examination - LOTCA Battery - Contextual Memory Test - Rey Complex Figure (copy) - Star cancellation task |
Motor perform. |
Orientation Repetition Pictorial sequence Geometrical seq. Visual memory Visual Attention |
ÃÆââ¬Å¡Ãâà0.40* 0.37*ÃÆââ¬Å¡Ãâà0.36* 0.36* 0.38* 0.53** |
||
| McDowd et al. 2003 (29) |
55 31 / 24 71.4 ± 6.4 39 control 17 / 22 72.6 ± 6.7 |
31 / 24 / 0 43.2 ± 18.6 months |
- Stroke Impact Scale |
- Attentional tasks |
Physical component of SIS1 | Divided attention Memory task1 Dual-single task1 Switching attention Cued Alternating |
ÃÆââ¬Å¡Ãâà0.30* 0.09 -0.11 -0.10 |
||
| Nas et al. 2004 (37) |
40 16 / 24 57.1 ± 12.9 |
16 / 24 / 0 138,9 ± 88.3 days |
- Functional Independence Measure (motor subscale) - Chedoke Assessment Impairment Inventory |
- Functional Independence Measure (cognitive subscale) |
Motor perform. (subscale FIM) |
Cognitive perform. (subscale FIM) |
Admission 0.47** Discharge 0.54** | ||
| Predicting motor outcome | |||||||||
| Carter et al. 1988 (39) |
21 12 / 9 68.6 ± 14.93 |
4 / 17 / 0 Not available |
- Revised Kenny Self-Care Evaluation |
- Cognitive skills evaluation |
Posttest locomotion | pretest auditory attention | ÃÆââ¬Å¡Ãâà0.49** | ||
| Heruti et al. 2002 (40) |
315 181 / 134 75.3 ± 7.6(60-94) |
150 / 139 / 26 3.96 ± 6.77 months (0-55) |
- Functional Independence Measure (motor subscale) |
- Functional independence Measure (cognitive subscale) - Mini Mental State Examination |
Motor gain Motor gain | Cognitive perform. (subscale FIM) MMSE |
ÃÆââ¬Å¡Ãâà0.48** 0.46** | ||
| Ones et al. 2009 (41) |
88 50 / 38 63.1 ± 10.1 |
53 / 35 / 0 9.3 ± 2.4 months |
- Functional Independence Measure (motor subscale) - Asworth scale - Brunnstrom Motor Evaluation Scale |
- Functional Independence Measure (cognitive subscale) - Mini Mental State Examination |
Motor perform. At discharge |
Cognitive perform (subscale FIM) MMSE |
ÃÆââ¬Å¡Ãâà0.51** 0.69** | ||
| Pahlman et al. 2011 (25) |
74 36 / 38 78 ± 8 (65-97) |
Not available 10.4 ± 9 days (0-38) |
- Berg Balance Scale |
- Cognitive Impairment Questionnaire - Neuropsychological test battery |
Post acute Balance | Deductive reasoning And EF |
ÃÆââ¬Å¡Ãâàn.s.* | ||
* p < 0.05, ** p <0.01, n.s.* correlation and/or p-value not specified but denoted as significant in results section, n.s. correlation and/or p-value not specified but denoted as non-significant in results section, † Correlation and/or p-value not specified, no information denoted on significance
Motor and cognitive impairment assessed at the same point in time
In 16 papers the link between motor and cognitive function was explored at the same time point in the same stroke patients.
Gait
Positive correlations between objective measures of basic gait and cognitive flexibility were found, while no correlation with working memory was found [31,32]. More complex gait tasks, with additional demands on EF, were correlated to response inhibition, planning and behaviour regulation [31]. Gait velocity (walking at normal pace) did not correlate with the MMSE [28] or with more specific functions, including cognitive flexibility or working memory [32]. Locomotion, evaluated by a motor rating scale, and the cognitive component comprehension were found to correlate at 4 points in time in one study. The link with other cognitive measures, for instance memory, calculation or construction, was less stable over time [33].
Balance
In 4 studies balance and cognitive functioning were correlated, using the Berg Balance Scale [22,23,32,34]. The link with specific cognitive domains was assessed and correlations with cognitive flexibility [32], sustained and divided attention [22], auditory selective attention [23,34] and visual selective attention [23] were found. The Single Leg Stance Test, an objective measure, failed to correlate with the MMSE [28].
Limb function
Impairment in limb function in relation to cognitive functioning was the subject of 4 studies [23,26,27,35] and a correlation with both attention and visuospatial functioning was found. Limb function, assessed by items on a rating scale which also included whole body movements, was correlated to sustained, divided and visual selective attention [23]. Furthermore, in right hemisphere stroke a correlation between functioning of the affected arm and sustained attention, but not divided attention, was shown 2 years post stroke [26]. In a sample of the general stroke population relatively high correlations were found between arm and leg motor function and visuospatial construction [27]. In right hemisphere stroke visuospatial impairment was related to dexterity errors in the ipsilateral hand [35].
Subjective motor performance
In 6 studies motor functioning was evaluated using rating scales completed either by clinicians [24,30,36,37] or patients themselves [29]. The motor subscale of the Functional Independence Measure (FIM) was used in a longitudinal study and a correlation with visuomotor organization was found at consecutive points in time [36] in populations with both neglect and non-neglect. In a sample of stroke patients without neglect, a correlation between the motor and cognitive subscale of the FIM was found [37]. The remaining studies used other motor rating scales, and found low to moderate correlations with several cognitive measures, including visual attention [38], while on the other hand no correlation was found with visual perception [24,30]. Finally a significant correlation between physical functioning, as evaluated with a self-report functional outcome measure, and divided attention was reported [29].
Motor and cognitive impairment in predicting motor functioning
5 studies focused on change in motor function over time and all showed that higher cognitive status at admission predicted better motor outcome [25,26,39-41]. In evaluating motor performance rating scales predominated, while cognitive functioning was assessed with either a global cognitive measure (MMSE) [40,41] or tasks measuring specific cognitive domains [25,26,39]. Correlations between baseline attention and locomotion [39] or dexterity in the affected hand [26] in the post acute timeframe, as well as baseline deductive reasoning and EF and improvement of balance [25] were found.
Discussion
The relation between motor and cognitive impairment after stroke within the same patients and using separate (objective) tasks or (subjective) rating scales for each domain was explored in the current review. Five of the included studies focused on predicting motor outcome and consistently showed that higher cognitive status in the acute phase of stroke predicted better motor outcome in the longer term. More importantly, we found a consistent correlation across studies between impairment in balance and limb function and deficits in selective, sustained and divided attention and also between gait and balance and cognitive flexibility. These correlations were found both in the acute and post-acute phase of stroke. The strength of the correlations appeared to depend on the type(s) of instruments used. They were more robust for objective measures assessing specific functions and weaker or more heterogeneous when subjective (rating scales) or global (screening) measures were used.
Attention and EF were the focus across studies when specific cognitive domains (rather than global screening instruments) were assessed. The link between various aspects of motor impairment and specific attention deficits has been consistently shown in stroke [22,23,26,29,34]. Findings for a link between motor functioning and EF have been less convincing: both balance and gait correlated with cognitive flexibility, but not with working memory [31,32]. A link between gait and planning and behavior regulation was shown [31], but this concerned complex gait tasks, involving additional cognitive load which was executive in nature. The inconsistency of results might be either due to the lack of a unified concept of EF, because EF encompasses too many different aspects to measure them all within the same patients or the possibility that, as shown in a meta-analysis [42], different executive tasks rely on different neural substrates suggesting that a broad neural network is likely for EF. In two papers motor functioning was not related to working memory [31,32]. However, in both papers the same cognitive task (digits backwards) was used, limiting the generalizability of the results. Furthermore, the link between motor functioning and other cognitive domains, for example memory or language has not yet been evaluated.
Our systematic review has several limitations. Overall, a small number of papers met the selection criteria and details about severity or other clinical variables were often lacking. In addition, in- and exclusion criteria varied between papers, in 3 papers a minimum score on the MMSE was required to include patients (range 18-24 items correctly answered), while in 3 other papers a modified MMSE or an alternative cognitive screening task was applied, reducing the range of cognitive impairment in these samples. Furthermore, methodological differences between studies were widespread making comparison between studies difficult. Finally, all papers included in this review used correlational analyses to link motor and cognitive functioning within the same patients, and although links were found (see above), no causal inferences can be made.
More research is clearly needed in order to investigate the specific links at both the behavioral and neural levels between motor deficit and cognitive impairment in stroke. Our review suggests that specific objective measures for both domains (motor and cognitive; i.e. not global screenings) might be more fruitful for future research than subjective measures. A broader range of cognitive domains, not just attention and EF should also be assessed.
Conclusion
In conclusion, motor and cognitive impairment after stroke were correlated, despite the heterogeneity in populations, designs and tasks used in the studies included in this review. The most consistent finding was that disturbances of gait, balance and limb function were often accompanied by deficits in attention and EF. Objective measures for specific motor and cognitive functions were also more valuable in finding this link than subjective measures. Unfortunately, subjective measures are more commonly employed in clinical practice, perhaps due to the fact that many of these measures can be quickly achieved. Our review suggests however that: a.) objective measures of both motor and cognitive performance might be more fruitful in finding a link between motor and cognitive impairment and b.) if specific aspects of one domain are impaired then we recommend also assessing the other domain. Both cognitive and motor impairment should be taken in to account when assessing the likelihood of rehabilitation success in stroke. Motor impairment (especially in the acute phase post stroke) might be more obvious to patients and carers. However, this review suggests that there may be underlying cognitive impairment too and that both domains should be assessed and evaluated before the patient begins their treatment plan.
References
- Planton M, Peiffer S, Albucher JF, Barbeau EJ, Tardy J, et al. (2012) Neuropsychological outcome after a first symptomatic ischaemic stroke with 'good recovery'. Eur J Neurol 19:212-219.
- Rasquin SM, Lodder J, Ponds RW, Winkens I, Jolles J, et al. (2004) Cognitive functioning after stroke: a one-year follow-up study. Dement Geriatr Cogn Disord 18:138-144.
- Jaillard A, Naegele B, Trabucco-Miguel S, LeBas JF, Hommel M (2009) Hidden dysfunctioning in subacute stroke. Stroke 40:2473-2479.
- Nys GM, van Zandvoort MJ, de Kort PL, van der Worp HB, Jansen BP, et al. (2005) The prognostic value of domain-specific cognitive abilities in acute first-ever stroke. Neurology 64:821-827.
- Lesniak M, Bak T, Czepiel W, Seniow J, Czlonkowska A (2008) Frequency and prognostic value of cognitive disorders in stroke patients. Dement Geriatr Cogn Disord 26:356-363.
- Hommel M, Miguel ST, Naegele B, Gonnet N, Jaillard A (2009) Cognitive determinants of social functioning after a first ever mild to moderate stroke at vocational age. J Neurol Neurosurg Psychiatry 80:876-880.
- Hommel M, Trabucco-Miguel S, Joray S, Naegele B, Gonnet N, et al. (2009) Social dysfunctioning after mild to moderate first-ever stroke at vocational age. J Neurol Neurosurg Psychiatry 80:371-375.
- Achten D, Visser-Meily JM, Post MW, Schepers VP (2012) Life satisfaction of couples 3 years after stroke. Disabil Rehabil 34:1468-1472.
- Carod-Artal FJ, Medeiros MS, Horan TA, Braga LW (2005) Predictive factors of functional gain in long-term stroke survivors admitted to a rehabilitation programme. Brain Inj 19:667-673.
- Barker-Collo S, Feigin V (2006) The impact of neuropsychological deficits on functional stroke outcomes. Neuropsychol Rev 16:53-64.
- Ozdemir F, Birtane M, Tabatabaei R, Ekuklu G, Kokino S (2001) Cognitive evaluation and functional outcome after stroke. Am J Phys Med Rehabil 80:410-415.
- Wassenberg R, Feron FJ, Kessels AG, Hendriksen JG, Kalff AC, et al. (2005) Relation between cognitive and motor performance in 5- to 6-year-old children: results from a large-scale cross-sectional study. Child Dev 76:1092-1103.
- Rigoli D, Piek JP, Kane R, Oosterlaan J (2012) An examination of the relationship between motor coordination and executive functions in adolescents. Dev Med Child Neurol 54:1025-1031.
- Hausdorff JM, Yogev G, Springer S, Simon ES, Giladi N (2005) Walking is more like catching than tapping: gait in the elderly as a complex cognitive task. Exp Brain Res 164:541-548.
- Ble A, Volpato S, Zuliani G, Guralnik JM, Bandinelli S, et al. (2005) Executive function correlates with walking speed in older persons: the InCHIANTI study. J Am Geriatr Soc 53:410-415.
- Taekema DG, Gussekloo J, Maier AB, Westendorp RG, de Craen AJ (2010) Handgrip strength as a predictor of functional, psychological and social health. A prospective population-based study among the oldest old. Age Ageing 39:331-337.
- Domellof ME, Elgh E, Forsgren L (2011) The relation between cognition and motor dysfunction in drug-naive newly diagnosed patients with Parkinson's disease. Mov Disord 26:2183-2189.
- Benedict RH, Holtzer R, Motl RW, Foley FW, Kaur S, et al. (2011) Upper and Lower Extremity Motor Function and Cognitive Impairment in Multiple Sclerosis. J Int Neuropsychol Soc 13:1-11.
- Scherder E, Eggermont L, Swaab D, van Heuvelen M, Kamsma Y, et al. (2007) Gait in ageing and associated dementias; its relationship with cognition. Neurosci Biobehav Rev 31:485-497.
- Yogev-Seligmann G, Hausdorff JM, Giladi N (2008) The role of executive function and attention in gait. Mov Disord 23: 329-342.
- Allali G, Van Der MM, Assal F (2010) Gait and cognition: The impact of executive function. Schweiz Arch Neurol Psychiatr 161:195-199.
- Hyndman D, Ashburn A (2003) People with stroke living in the community: Attention deficits, balance, ADL ability and falls. Disabil Rehabil 25:817-822.
- Hyndman D, Pickering RM, Ashburn A (2008) The influence of attention deficits on functional recovery post stroke during the first 12 months after discharge from hospital. J Neurol Neurosurg Psychiatry 79:656-663.
- Bernspang B, Asplund K, Eriksson S, Fugl-Meyer AR (1987) Motor and perceptual impairments in acute stroke patients: effects on self-care ability. Stroke 18:1081-1086.
- Pahlman U, Gutierrez-Perez C, Savborg M, Knopp E, Tarkowski E (2011) Cognitive function and improvement of balance after stroke in elderly people: the Gothenburg cognitive stroke study in the elderly. Disabil Rehabil 33:1952-1962.
- Robertson IH, Ridgeway V, Greenfield E, Parr A (1997) Motor recovery after stroke depends on intact sustained attention: a 2-year follow-up study. Neuropsychology 11:290-295.
- Sveen U, Bautz-Holter E, Sødring KM, Wyller TB, Laake K (1999) Association between impairments, self-care ability and social activities 1 year after stroke. Disabil Rehabil 21:372-377.
- Taylor-Piliae RE, Latt LD, Hepworth JT, Coull BM (2012) Predictors of gait velocity among community-dwelling stroke survivors. Gait Posture 35:395-399.
- McDowd JM, Filion DL, Pohl PS, Richards LG, Stiers W (2003) Attentional abilities and functional outcomes following stroke. J Gerontol B Psychol Sci Soc Sci 58:P45-53.
- Deshpande LA, Rege RV (1999) Study of impact of cognitive perceptual deficits on daily living in adults with brain damage. Indian J Occup Environ Med 3:162-165.
- Hayes S, Donnellan C, Stokes E (2013) Associations between executive function and physical function poststroke: a pilot study. Physiotherapy 99:165-171.
- Liu-Ambrose T, Pang MY, Eng JJ (2007) Executive function is independently associated with performances of balance and mobility in community-dwelling older adults after mild stroke: implications for falls prevention. Cerebrovasc Dis 23:203-210.
- Fong KN, Chan CC, Au DK (2001) Relationship of motor and cognitive abilities to functional performance in stroke rehabilitation. Brain Injury 15:443-453.
- Stapleton T, Ashburn A, Stack E (2001) A pilot study of attention deficits, balance control and falls in the subacute stage following stroke. Clin Rehabil 15:437-444.
- Sunderland A, Bowers MP, Sluman SM, Wilcock DJ, Ardron ME (1999) Impaired dexterity of the ipsilateral hand after stroke and the relationship to cognitive deficit. Stroke 30:949-955.
- Katz N, Hartman-Maeir A, Ring H, Soroker N (2000) Relationships of cognitive performance and daily function of clients following right hemisphere stroke: Predictive and ecological validity of the LOTCA battery. Occupational Therapy Journal of Research 20:3-17.
- Nas K, Gur A, Cevik R, Sarac AJ (2004) The relationship between physical impairment and disability during stroke rehabilitation: effect of cognitive status. Int J Rehabil Res 27:181-184.
- Kizony R, Katz N (2002) Relationships between cognitive abilities and the process scale and skills of the Assessment of Motor and Process Skills (AMPS) in patients with stroke. OTJR Occup Part Health 22:82-92.
- Carter LT, Oliveira DO, Duponte J, Lynch SV (1988) The relationship of cognitive skills performance to activities of daily living in stroke patients. Am J Occup Ther 42:449-455.
- Heruti RJ, Lusky A, Dankner R, Ring H, Dolgopiat M, et al. (2002) Rehabilitation outcome of elderly patients after a first stroke: effect of cognitive status at admission on the functional outcome. Arch Phys Med Rehabil 83:742-749.
- Ones K, Yalcinkaya EY, Toklu BC, Caglar N (2009) Effects of age, gender, and cognitive, functional and motor status on functional outcomes of stroke rehabilitation. NeuroRehabilitation 25:241-249.
- Niendam TA, Laird AR, Ray KL, Dean YM, Glahn DC, et al. (2012) Meta-analytic evidence for a superordinate cognitive control network subserving diverse executive functions. Cogn Affect Behav Neurosci 12:241-268.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences